- Blog
- Platypus evolution game online
- Prasanth kishore
- Companionlink invalid class string
- Toon boom animate pro 2 full download
- Japji sahib song
- Genius only you cheat codes
- Yu yu hakusho opening 2 start
- Coreldraw x7 update
- Block adobe hosts 2019
- Simpleware scan ip tutorial and reference guide
- Synergy 1-8-8 ubuntu
- Background music get busy sean paul
- Disk drill 3 activation code
- Sony vaio s 15 2012 review
- Free poser and daz models
- Bridgit mendler ready or not album cover
- Manowar warriors of the world album cover
- Bleeding love sytycd
- What year did 2pac die
- Gta san andreas pc
- Ableton live 10 crack
- Kelk 2010 patcher v3
- Minecraft more fps mod 1-14-3
- Sonnox oxford bundle
- Old bollywood movies hd
- Ellie kemper hot kissing scene
- Blog
- Platypus evolution game online
- Prasanth kishore
- Companionlink invalid class string
- Toon boom animate pro 2 full download
- Japji sahib song
- Genius only you cheat codes
- Yu yu hakusho opening 2 start
- Coreldraw x7 update
- Block adobe hosts 2019
- Simpleware scan ip tutorial and reference guide
- Synergy 1-8-8 ubuntu
- Background music get busy sean paul
- Disk drill 3 activation code
- Sony vaio s 15 2012 review
- Free poser and daz models
- Bridgit mendler ready or not album cover
- Manowar warriors of the world album cover
- Bleeding love sytycd
- What year did 2pac die
- Gta san andreas pc
- Ableton live 10 crack
- Kelk 2010 patcher v3
- Minecraft more fps mod 1-14-3
- Sonnox oxford bundle
- Old bollywood movies hd
- Ellie kemper hot kissing scene
16, 17 Despite its importance for quantitative fNIRS analysis, the available mesh-based brain models remain limited 6, 18, 19 in part due to the difficulties in generating accurate brain tetrahedral meshes. 12, 13 In fNIRS, tetrahedral meshes have been reported in several studies to model light propagation and recover brain hemodynamic activation using the finite-element 14, 15 or mesh-based Monte Carlo (MMC) method. Mesh-based models are not only the most common choice in computer graphics and 3-D visualization of brain structures, but they are also the primary format for finite element analysis (FEA) and image reconstructions in many neuroimaging studies.

Mesh-based brain/head models made of triangular surfaces or tetrahedral elements have advantages in both improved boundary accuracy and high flexibility compared to voxelated domains. This improves memory efficiency significantly but still suffers from terraced mesh boundaries. Another approach-octree-uses nested voxel refinement near curved boundaries. This can cause prohibitive memory allocation and runtime in applications where solving sophisticated numerical models is necessary. In addition, the uniform grid structure of the voxel space also demands a large number of cells in order to store brain anatomy without losing spatial details. Whereas a voxelated brain representation has been dominantly used for acquiring and storing a three-dimensional (3-D) neuroanatomical volume, the terraced boundary shape in a voxelated space has difficulty in representing a smooth and curved boundary that typically delineates human tissues, resulting in a loss of accuracy, especially when modeling complex cortical surfaces with limited resolution. 3, 6 In addition, modeling brain anatomy accurately also plays important roles in other quantitative neuroimaging modalities, such as electroencephalography (EEG) 7 and magnetoencephalography. While much simplified brain models, such as planar 2, 3 or spherical layers, 4 as well as approximated photon propagation models, such as the diffusion approximation (DA), 5 have been widely utilized by the research community, their limitations are recognized by a number of studies. Using the anatomical scans of the patient’s head or a resembling atlas, Monte Carlo (MC) simulations are often used in conjunction with tissue optical properties to approximate the photon pathlengths that are used to reconstruct the changes in HbR and HbO 2.
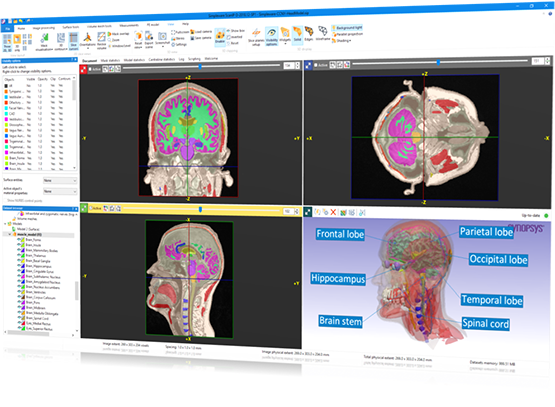
The accuracy of this inference depends greatly not only upon an accurate representation of the complex human brain anatomy but also on the surrounding tissues that affect the migrations of photons from the sources to the detectors. Relative changes in oxygenated ( HbO 2) and deoxygenated hemoglobin (HbR) concentrations, as a result of neural activities, lead to variations in light intensities at the detectors that are used to infer the locations of these activities. 1 Using light in the red and near-infrared range, the hemodynamic response of the brain is probed through careful placement of sources and detectors on the scalp surface at multiple wavelengths. įunctional near-infrared spectroscopy (fNIRS) has played an increasingly important role in functional neuroimaging. Our open-source meshing toolboxes “Brain2Mesh” and “Iso2Mesh” are freely available at.

In addition, we compare three brain anatomical models-the voxel-based brain segmentation, tetrahedral brain mesh, and layered-slab brain model-and demonstrate noticeable discrepancies in brain partial pathlengths when using approximated brain anatomies, ranging between −1.5 % to 23% with the voxelated brain and 36% to 166% with the layered-slab brain.Ĭonclusion: The generation and utility of high-quality brain meshes can lead to more accurate brain quantification in fNIRS studies. Results: A variety of high-quality brain mesh models have been successfully generated by processing publicly available brain atlases. It can convert segmented volumetric brain scans into multilayered surfaces and tetrahedral mesh models, with typical processing times of only a few minutes and broad utilities, such as in Monte Carlo or finite-element-based photon simulations for fNIRS studies. Accurate quantification of brain activities via fNIRS relies upon solving computational models that simulate the transport of photons through complex anatomy.Īim: We aim to highlight the importance of accurate anatomical modeling in the context of fNIRS and propose a robust method for creating high-quality brain/full-head tetrahedral mesh models for neuroimaging analysis.Īpproach: We have developed a surface-based brain meshing pipeline that can produce significantly better brain mesh models, compared to conventional meshing techniques. Significance: Functional near-infrared spectroscopy (fNIRS) has become an important research tool in studying human brains.
